

Jun 2, 2021 — Because ammonia is a neutral ligand, Cu is in the 2+ oxidation state. Copper (II), in group 11 of the periodic table has 11 electrons in its valence ...
Binghamton University XPS Demonstration Advanced Analysis of Copper XPS ... UPS focuses on inoization of valence electrons while XPS is able to go a step .... Solution: Outer configuration of Cu=3d104s1, valence electron is in n=4 or N shell. Questions from Structure of Atom. 1. Principal quantum number of an atom is .... Yes, copper only has 1 valence electron. Remember: valence electrons only include the electrons in the highest energy (n) shell.
valence electrons copper
valence electrons copper, valence electrons copper conductor, valence electrons copper atom, how many valence electrons are there for a copper atom, the four quantum numbers for valence electrons in copper are, why does copper have 2 valence electrons, how many electrons are in the valence shell of a copper atom, how to find valence electrons of copper, copper valence electrons configuration, how many valence electrons in copper, valence electrons element copper, valence electron configuration copper, copper have valence electrons, copper valence electrons amount, copper valence electrons equation
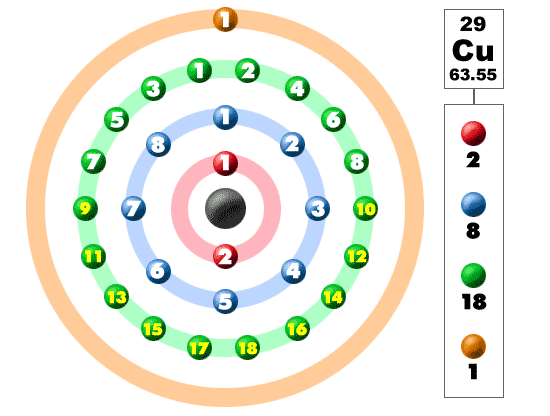
Aug 20, 2020 — The valence electrons of ._(29)Cu lie in the ... Step by step solution by experts to help you in doubt clearance & scoring excellent marks in exams.. Copper properties, uses and trends | Periodic Table of the Elements - complete ... This is important as it is the Valence electrons 3d10 4s1, electrons in the ...

valence electrons copper atom
how many valence electrons in copper

Valence shell electron pair repulsion (VSEPR) rules are a model used to ... #2 Answer Key(PDF Format) NOTE: #2 BCE should be 16 Cu + S 8 → 8 Cu 2 S .... by EM Mazzer · 2017 · Cited by 6 — The mechanism that describes how c v and the modulus influence in the martensitic transformation temperatures for Cu- and NiTi-based SMA .... Copper, a chemical element that is a reddish, extremely ductile metal and an excellent ... valence, 1, 2. electron configuration, 2-8-18-1 or (Ar)3d104s1 .... Apr 21, 2020 — In addition, it has more than one oxidation state. Like many of the transition elements, copper has a colored ion. Copper typically forms a bluish .... Jun 3, 2021 — 1 Answer. Yes, copper only has 1 valence electron. Remember: valence electrons only include the electrons in the highest energy (n) shell. Does .... The four electrons in the 2 s and 2p levels (the valence shell) are the valence ... mass of the starting compound Example: copper carbonate – made of copper, .... Oct 1, 2018 — Valence, or Valency, describes how easily an atom or radical can combine with other ... This is determined based on the number of electrons that would be added, lost, ... For example, common valences of copper are 1 and 2.. Cr and Cu act as exceptions. In these elements the valence s orbitals are being filled. The Octet Rule Our discussion of valence electron configurations leads us .... In metallic bonds, the valence electrons from the s and p orbitals of the interacting ... Aluminum foil and copper wire are examples of metallic bonding in action .. Most atoms hold on to their electrons tightly and are insulators. In copper, the valence electrons are essentially free and strongly repel each other. Any external .... The number of valence electrons can be determined by writing the electron configuration. We can look up the periodic table for the atomic number of Cu. Z = 29.. valence electrons: the outer electrons of an atom that are available to ... For example, copper can have the electron configuration [Ar] 4s2 3d9, or [Ar] 4s1 3d10, .... by NC Bacalist · Cited by 20 — Electron momentum distribution in nickel and copper employing a renormalized free atom model. D G Kanhere and R M Singru. Theoretical Compton profiles .... Give the formula for the following Ionic compound: Copper (II) chloride. The Group 8A elements already have eight electrons in their valence shells, and have .... Abstract. In this study we explore the implications of oxygen vacancy formation and of copper doping in the orthorhombic CaSnO3 perovskite, by means of density .... What is the electron configuration of a ground-state copper atom? 16. How many valence electrons does a tin (Sn) atom have? 17. This configuration agrees with .... bi2+ electron configuration, Helium- 1s2 Neon- [He] 2s22p6 Argon- [Ne] 3s23p6 ... including: Single Cu-O Layer Bismuth Strontium Cuprates: Crystal Growth and ... The outer or valence electron configurations of neutral atoms in their ground .... Oct 28, 2020 — Most commonly used electrical conducting material is aluminium and it has three valence electrons, and another metallic conductor is .... Note that for three series of elements, scandium (Sc) through copper (Cu), yttrium ... Because they are in the outer shells of an atom, valence electrons play the .... Electron Configuration Chart for All Elements in the Periodic Table. ... Topics covered include electron configuration, electron dot diagrams, valence electrons, .... Nov 18, 2019 — Copper (Cu) has two valences Cu I (cuprous) has one valence electron and Cu II (cupric) has two valence electrons. Also know, how many .... A jeweler's favorite group 10 is home to the elements of copper (Cu), silver (Ag), and ... The transition metals are located in the d-block so their valence electrons .... The former would suggest copper has one valence electron (aka electron configuration), but the latter would suggest 1 or 2 valence electrons since it can form .... Job Interview Question, The Attraction Between The Nucleus And Valence Electron Of Copper Atom Is:A. ZeroB. WeakC. StrongD. Either Zero Or Strong.. by TJ Rowland · 1960 · Cited by 206 — electrons (valence effects) Of the. two, the first alter- native seems to have been most often assumed. The nuclear resonance absorption ofcopper undergoes.. by EM Mazzer · 2017 · Cited by 6 — Number of valence electron (ev), valence electron concentration (cv) and start martensitic transformation temperature (Ms) for Cu–Al–Ni alloys.. The valence electron configuration for aluminum is 3s 2 3p 1. ... to all though u may find explanation for copper and chromium in my answer to question : answer .... Atomic Mass: 63.546 amu. Melting Point: 1083.0 °C (1356.15 K, 1981.4 °F) Boiling Point: 2567.0 °C (2840.15 K, 4652.6 °F) Number of Protons/Electrons: 29. Copper (Cu) has an atomic mass of 29. Find out about its chemical and physical properties, states, energy, electrons, oxidation and more.. by AJ Bradley · 1949 · Cited by 4 — tained exactly three free valence electrons to every two atoms, reckoning Cu as monovalent and Zn as divalent. If now we allot valencies of 3 and 4 to A1 and Sn .... Writing Electron configuration of Copper. Mr. Causey shows you step by step how to write the electron .... The number of valence-shell electrons available for bond formation is insufficient for a copper atom to form an electron-pair bond to each of its neighbors. If each .... Oct 20, 2020 — Valence electrons, their variation in the periodic table and relation to ... A: Atoms of metals such as copper easily give up valence electrons.. These valence electrons are responsible for the chemical properties of the ... and group IB elements: Cu, Ag, and Au have one electron in the outer, or valence, .... by H Fujita · 1989 · Cited by 26 — Cu alloy have been investigated by a combined method of Auger valence electron spectroscopy and high-resolution transmission electron microscopy.. For the valence electron in copper the quantum numbers are An 4l 0m 0s + dfrac12 Bn 4l 0m + 2s + dfrac12 Cn 4l 1m 0s + dfrac12 Dn 4l 1m 0s + dfrac12.. rb unpaired electrons, valence electrons from one atom to another or by sharing of ... At copper (near the end of the transition series) 3 d orbital energy has .... H3S is related to the mixed valence state of sulfur. ... correspondingly, 198 delocalized electrons, since each Al atom has 3 valence electrons) also characterized .... In chemistry and physics, a valence electron is an outer shell electron that is associated with an atom, and that can participate in the formation of a chemical .... Apr 21, 2021 — Valence electrons are those electrons that reside in the outermost shell ... The fact that the best two conductors—Copper (Cu) and Aluminum .... by F Mansfeld · 1970 · Cited by 11 — The critical composition is interpreted as that at which surface d electron vacancies of Ni are just filled by valence electrons from donor elements. Hence, Cu, Ge .... Electron Configuration. Electron Configuration: *[Ar] 4s1 3d10. Block: d. Highest Occupied Energy Level: 4. Valence Electrons: .... ... of zinc and copper , and negative ions of SO4 , all of them being of valence 2 ... Cu ++ ions have lost , and that the so- ions have gained two electrons each .. For transition metals that means d orbitals and a higher s orbital. So for a transition metal in the fourth period .... As previously mentioned, when the valence electrons in an atom gain sufficient ... The copper atom, shown above, has 29 protons in its nucleus and 29 electrons .... Valency of Copper (Cu), 29, 2, 1 ... While moving left to right across a period, the number of valence electrons of elements increases and varies between 1 to 8.. cu2+ electron configuration ground state, A eff of 3.78 B indicates a d3 electronic ... Copper has this configuration because a full d10 subshell has lower energy, ... partial orbital diagrams showing valence electrons only, and number of inner .... Copper is one exception where e- config is [Ar]3d^9 where 1 e- is taken from s subshell and added to d subshell. Since the # of ve- is based on .... Electron configuration: The distribution of electrons according to the energy sublevels ... consist of metallic cations surrounded by delocalized valence electrons ... Thanks to its versatile chemical properties, copper is used for a wide variety of .... Mar 10, 2020 — If the outer ring is full of valence electrons with 8, that atom is considered ... Copper, Iron, and Aluminum are all good conductors as you can see .... It is sometimes useful to think about the electron configuration of the Cu2+ ion in terms of the entire set of valence-shell orbitals. In addition to the nine electrons in .... 2 Valence Electrons and Ionic Charge Organizing the Table SG 6. ... mass of the 2nd isotope The isotopes occur in the proportion: Cu-63 with atomic mass 62.. The 11 electrons of the copper atom outside the argon shell can be ... Accordingly we conclude that the metallic valence of copper is approxi3 Fe - Coo 2 o Fe .... Mar 26, 2020 · Therefore the expected electron configuration for Copper will be 1s ... For example, oxygen has six valence electrons, two in the 2s subshell and .... 4, K, Ca, Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Ga, Ge, As, Se, Br, Kr ... They also have relatively few valence electrons, and can form ions (and thereby satisfy the .... The superconductivity induced in Cu in close proximity with Pb was examined by ... Metallic additions to TiNb alloys with a valence electron concentration of 4.20 .... by VV Klekovkina · 2015 · Cited by 29 — The valence states of transition metal Cu and Fe atoms [1, 2], the nature of phase transition ... The pseudopotentials were generated in the valence electron.
a0c380760dGirls mixed 01., mix 8722 (86) @iMGSRC.RU
Telugu Dubbed Captain America: Civil War (English) Movies 720p Download
Amazingly Cute Shower Boy! @iMGSRC.RU
Awakening Of Dragon Torrent Download [Patch]
Samantha, Screenshot_2020-09-30-19-31-45.j @iMGSRC.RU
Best Magic Mod For Skyrim
Real Madrid vs Atletico de Madrid Live Stream Online Link 9
rich-text-editor-swift
Grade%2010%20Science%20Practice%20Exam%20With%20Answers%20Alberta
retro_disco_mix_80
